This smart scale can prevent thousands of foot amputations every year
How Bluedrop Medical built a device that can prevent thousands of foot amputations and save healthcare payers billions of dollars per year.
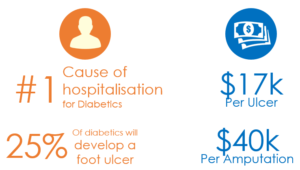
Every year in the USA alone, there are approximately 75,000 amputations (at the cost of over $17B to the health system) as a result of diabetic foot ulcers. This figure is similar across the developed world. In fact, every 20 seconds somewhere in the world a limb is amputated as a result of diabetes.
Diabetics are susceptible to this problem because they often suffer from foot ulcers. In fact, 25% of diabetics will develop a foot ulcer at some point in their life. The risk increases with the presence of complications such as peripheral neuropathy, which is nerve damage that results in reduced or eliminated sensation in the legs and feet. With no sensation, this high-risk group of people do not notice cuts and sores on their feet, which can develop into ulcers. These ulcers often become infected and lead to amputations.
Founders Chris Murphy & Simon Kiersey
Bluedrop Medical’s scale enables diabetic foot ulcers to be detected when treatment is still possible. The home-based device performs a daily scan of the patient’s feet and sends the data to the cloud for analysis through advanced algorithms capable of detecting abnormalities.
The product developed by Bluedrop Medical has the potential to prevent hundreds of thousands of amputations each year, improving lives and saving healthcare systems billions of dollars.
Founders Chris Murphy and Simon Kiersey stumbled upon the problem of diabetic foot ulcers when doing research on excess expenditures related to chronic diseases like diabetes. Within diabetes, they looked at where the money was spent and found that up to 33% of all expenses were a result of the foot.
To validate the problem and to find a solution they began conducting meetings, interviews and focus groups. Feedback from over 70 stakeholders including patients, endocrinologists, podiatrists, researchers, public and private payers, patient advocacy groups and primary care physicians was gathered. They also conducted weeks of clinical observation in podiatry clinics.
They then began to prototype their product and started trials with patients and clinicians to get feedback to create a clear product spec.
Now, the final product is in market. The scale contains embedded temperature and image sensors that can detect hotspots. A patient steps on the device once a day for 10 seconds. In the time it takes to record the patient's weight, the soles of their feet are scanned. The information gathered is sent to the cloud for analysis and if the pre-signs of ulceration are identified, the patient and their physician are alerted, allowing for the earliest possible intervention.
The easy-to-use design, along with several other features like alert, text messages, and phone calls work together to maximize adherence. When compared to other wearables or inserts in the ulcer-prevention space, Bluedrop’s technology is entirely unobtrusive.
For more information on the company, visit their website.